 The electrons determine the chemical properties of an element, and strongly influence an atom's magnetic properties. Electrons that are bound to atoms possess a set of stable energy levels, or orbitals, and can undergo transitions between them by absorbing or emitting photons that match the energy differences between the levels. This can result in a transmutation that changes the number of protons or neutrons in a nucleus. Each element has at least one isotope with unstable nuclei that can undergo radioactive decay. Over 99.9% of an atom's mass is concentrated in the nucleus, with protons and neutrons having roughly equal mass. Atoms can only be observed individually using special instruments such as the scanning tunneling microscope. Realizing that classical physics fails catastrophically when things are atom-sized or smaller, he remodeled the atom so electrons occupied ‘allowed’ orbits around the nucleus while all other orbits were forbidden. Relative to everyday experience, atoms are minuscule objects with proportionately tiny masses. Niels Bohr completely transformed our view of the atom and of the world. The principles of quantum mechanics were used to successfully model the atom. During the late 19th and early 20th centuries, physicists discovered subatomic components and structure inside the atom, thereby demonstrating that the 'atom' was divisible. In the 17th and 18th centuries, chemists provided a physical basis for this idea by showing that certain substances could not be further broken down by chemical methods. The concept of an atom as an indivisible component of matter was first proposed by early Indian and Greek philosophers. The name atom comes from the Greek ἄτομος/átomos, α-τεμνω, which means uncuttable, something that cannot be divided further. An atom is classified according to the number of protons and neutrons in its nucleus: the number of protons determines the chemical element, and the number of neutrons determine the isotope of the element. 
An atom containing an equal number of protons and electrons is electrically neutral, otherwise it has a positive or negative charge and is an ion. Likewise, a group of atoms can remain bound to each other, forming a molecule. The electrons of an atom are bound to the nucleus by the electromagnetic force.  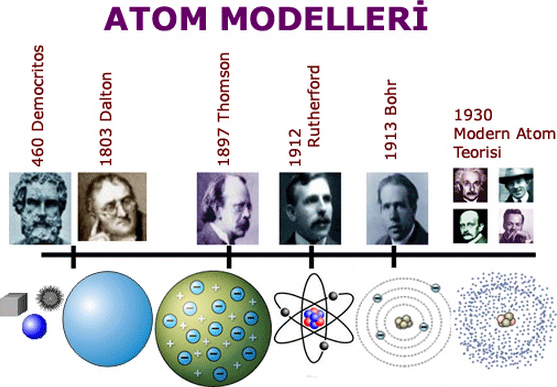
The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons (except in the case of hydrogen-1, which is the only stable nuclide with no neutron). The atom is a basic unit of matter consisting of a dense, central nucleus surrounded by a cloud of negatively charged electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
